Business Model
|
|
Long Term
|
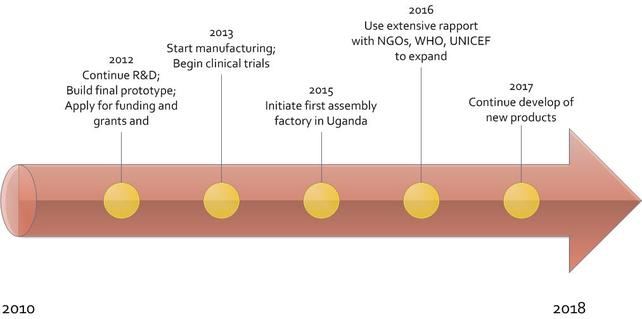
During Phase I, IncuVive plans to perform clinical testing after review from the Institutional Review Board (IRB) at Columbia University. Additional modifications will be made to the design when needed. Phase II will take place in 2013-2015 timeframe where IncuVive develops the final product and seek international approval. During this period, we will continue to run clinical tests at the Mulago Hospital. From 2015 to 2021, we will aggressively market (Phase III) our life-saving product to NGOs and government organizations in developing countries. Expansion will take place from 2016-2021, concurrently with Phase III. By this phase, we should be making a profit from our original device and should therefore have enough funds to contribute to the research and development of new devices. Furthermore, with the support of NGOs and charitable foundations, we will be able to market our product at a lower price. This will allow our Modular Incubation System to reach the needs of as many infants as possible.
|
|
Home | Company Overview | Products | Business Model | Downloads | About Us
4785 Lerner Hall, 2920 Broadway, New York, NY 10027 | [email protected] |
Copyright © 2011-2012 Kiet Vo | All rights reserved
|